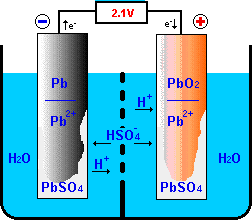
In the constant voltage charging method, charging voltage is kept constant throughout the charging process. In the constant current method, a fixed value of current in amperes is passed through the battery till it is fully charged. Electrical energy is converted into chemical energy which is stored in the cell.The terminal voltage of each battery cell increases to 2.2 to 2.5V.Water is consumed and sulphuric acid is formed which increases the specific gravity of electrolyte from 1.18 to 1.28.The anode is transformed into lead peroxide (PbO 2) and cathode into the spongy lead (Pb).Ultimately, the cell regains its previous (before discharging) chemical compositions. This increases the specific gravity of the electrolyte. Water (H 2O) is consumed and H 2SO 4 is formed in above chemical reactions. This electron is given by sulfate ion at the anode and has come to cathode via external circuit (from the DC source) 2H 2e -> 2H The hydrogen gas liberated at the cathode acts chemically on the cathode material (PbSO 4) PbSO 4 2H -> Pb H 2SO 4 Thus, PbSO 4 is transformed to Pb. The chemical reactions are as under: At anode SO 4 – – – 2e -> SO 4 (radical) SO 4 H 2O -> H 2SO 4 O The oxygen (in atomic stage) reacts chemically with anode material (PbSO 4): PbSO 4 O H 2O -> PbO 2 H 2SO 4 At cathode Each hydrogen ion (H ) on reaching the cathode, takes one electron from it to become hydrogen gas. During recharging, hydrogen ions (2H ) travel towards the cathode and sulfate ions (SO4 – –) travel towards the anode. 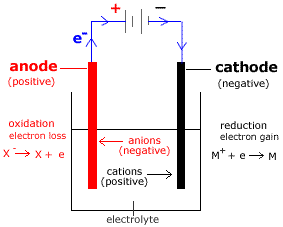
For recharging, positive terminal of DC source is connected to positive terminal of the battery (anode) and negative terminal of DC source is connected to the negative terminal (cathode) of the battery. The lead-acid battery can be recharged when it is fully discharged. Chemical energy is converted into electrical energy which is delivered to load.The terminal voltage of each battery cell falls to 1.8V.Sulphuric acid is consumed and water is formed which reduces the specific gravity of electrolyte from 1.28 to 1.18.Both the plates are transformed into lead sulfate (PbSO4).How do lead acid batteries work? Learn about the types of batteries used on ships PbO 2 2H -> PbO H 2O This PbO 2 is reduced to PbO which reacts with H 2SO 4 and forms PbSO 4. 2H 2e = 2H The hydrogen gas liberated at the anode acts chemically on the anode material (PbO 2). This electron is the same electron which is given by the sulfate ion at the cathode and has come to the anode via external load resistor R. The chemical reactions at the cathode are as under: At cathode SO4 – – – 2e = SO 4 (radical) Pb SO 4 -–> PbSO 4 At anode Each hydrogen ion (H ) on reaching the anode, takes one electron from it to become hydrogen gas. When the load resistance is connected to terminals of the battery the sulfate ions (SO4 – –) travel towards the cathode and hydrogen ions (2H ) travel towards the anode. When the sulphuric acid is dissolved, its molecules are dissociated into hydrogen ions (2H ) and sulfate ions (SO4 – –) which moves freely in the electrolyte. Charging and Discharging of Lead Acid Battery Discharging of Lead Acid Battery Whereas, when it is taking electrical energy from the external DC source, the process is known as charging of the cell. When the cell is delivering electrical energy to the external circuit (load), the process is known as discharging of the cell. The positive plate (anode) is made up of lead-peroxide (PbO 2) and the negative plate (cathode) is made up of sponge lead (Pb). A lead-acid cell basically contains two plates immersed in electrolyte (dilute sulphuric acid i.e. It consists of a number of lead-acid cells connected in series, parallel or series-parallel combination. A lead-acid battery is the most inexpensive battery and is widely used for commercial purposes. This ( ) sign stays the same in the charge and in the discharge processes.Hi friends, In this article, I am discussing about the charging and discharging of lead acid battery and hope you will find it informative and helpful. The lead acid battery has two electrodes, one made of metallic lead, and the other made of lead dioxide $\ce$ plate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
